 We also found that CMA becomes increasingly important for the maintenance of functional HSCs in aging, since as mice age, CMA-deficient HSCs showed an even greater functional defect compared to age-mated control-derived HSCs. We found reduced long-term colony formation of CMA-deficient HSCs in serial colony formation assays and demonstrated that these cells had a significant and progressive disadvantage of repopulating lethally-irradiated congenic recipient mice upon serial bone marrow transplantation. This upregulation of CMA is necessary to ensure HSCs persistence during activation because, upon serial 5-FU injections, CMA-deficient HSCs had a significantly reduced multilineage reconstitution ability with premature bone marrow failure. Our work revealed that the basal CMA activity detected in quiescent HSC under steady-state conditions is significantly stimulated upon HSC activation following 5-fluorouracil (5-FU) in vivo exposure. 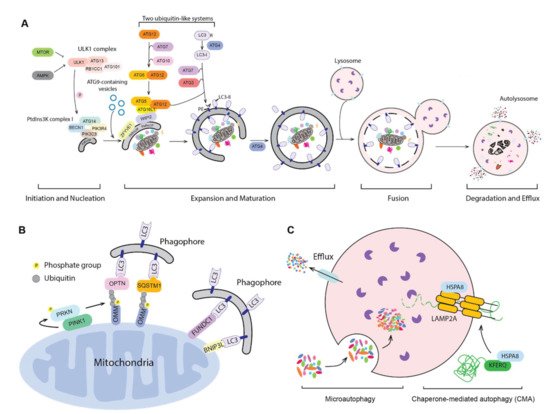 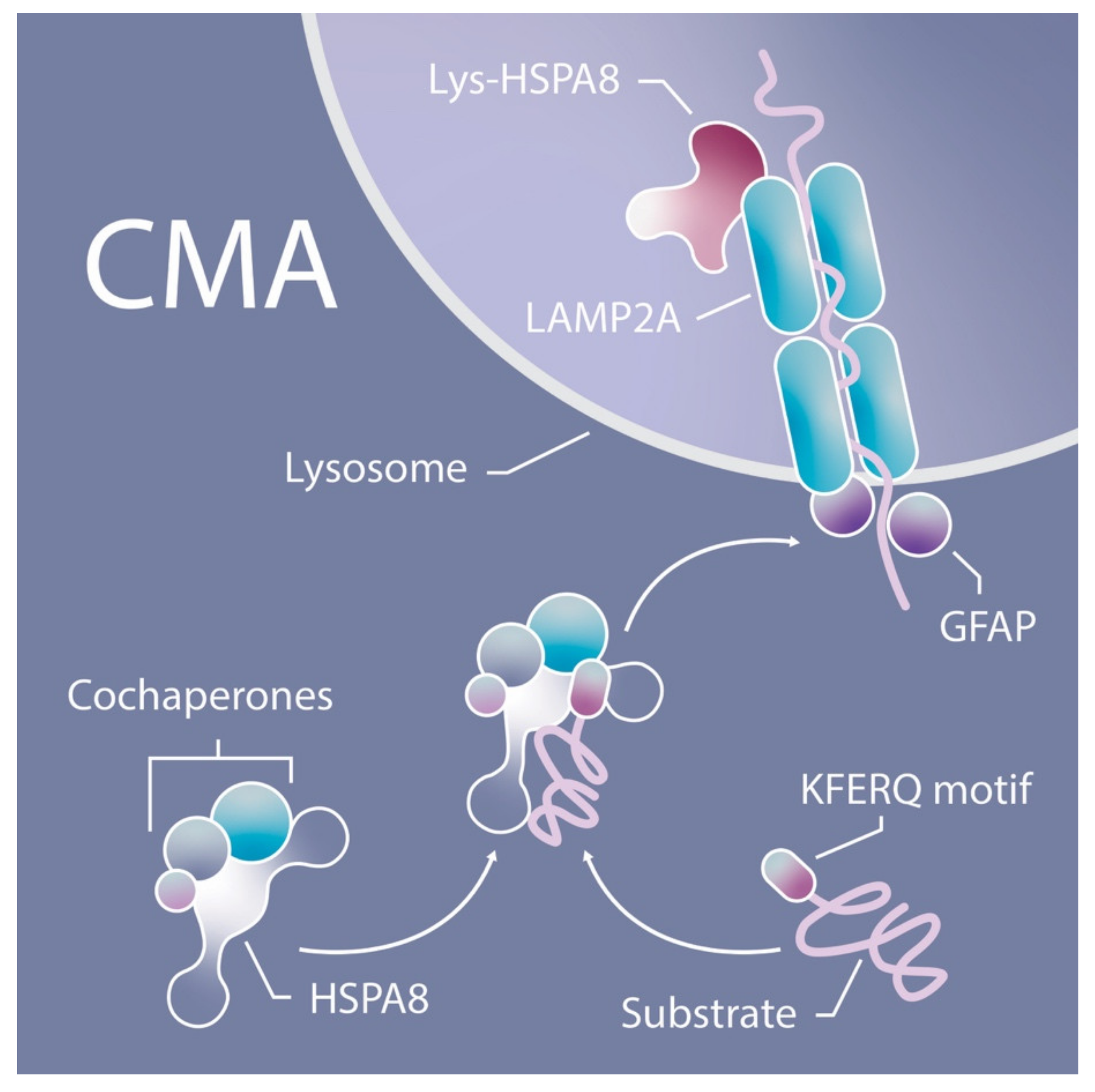
Using novel mouse models that allow for CMA tracking (KFERQ-Dendra mice) and for selective depletion of LAMP-2A in hematopoietic cells (Vav-iCre:LAMP2A f /f mice), we have investigated the physiological role of CMA in HSCs during steady-state and upon activation and gained novel insights on the consequences of CMA failure in these cells in aging. The main component of this complex is the lysosome-associated membrane protein type 2A (LAMP-2A) that also serves as substrate receptor and is the limiting component of CMA. Substrate proteins are directly translocated into the lysosomal lumen through a dedicated multiprotein translocation complex. In this work, we have focused in a highly selective type of autophagy, known as chaperone-mediated autophagy (CMA), whereby individual proteins bearing a unique pentapeptide motif (KFERQ-like) are targeted for degradation in lysosomes upon binding to the heat shock cognate protein of 70 kDa (HSC70). 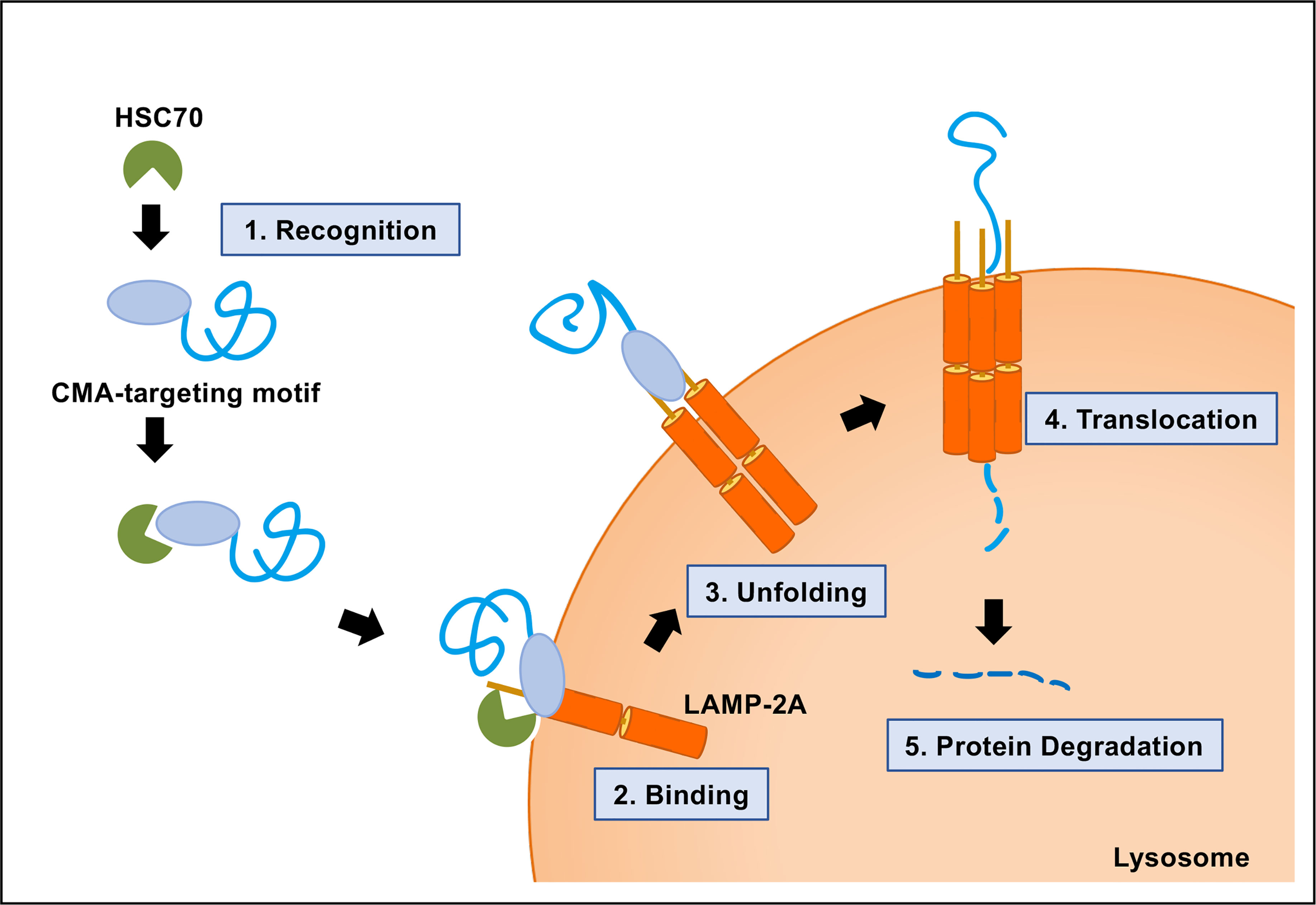
Alterations in HSC proteostasis have been associated with a number of degenerative and malignant diseases, underscoring the importance of elucidating the precise contribution of components of the cellular proteostasis network to HSC maintenance. Maintenance of a fully functional proteome that can rapidly change to adjust to the status of HSC activation is facilitated in part by two major intracellular proteolytic pathways, the ubiquitin proteasome system and the autophagy/lysosomal system. Hematopoietic stem cells (HSC) harbor extensive self-renewal capabilities along with multilineage differentiation plasticity to sustain blood production over a lifetime. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
